 How RSUs Can Impact Work Comp Premiums
How RSUs Can Impact Work Comp PremiumsTailor-Made Insurance for Small Businesses
Big Results for Small Businesses
We Take Care of the Risk so You can Take Care of your Growing Business
Heffernan Insurance Brokers is the go-to source for insurance for small businesses, from sole proprietors working from home to fast-growing companies with ever-changing needs. Our small-business division is part of our overall commitment to the insurance needs of companies of all sizes. Although we have grown into a $100 million company over the past four decades, we always remember that small business drives our economy, as well as our own success.
The types of coverage we can provide include (but are not limited to):
- General liability insurance covers bodily injury and property damage caused by your company, by both people and products.
- Workers’ compensation covers injuries to your employees while on the job.
- Disability provides income to your employees if they are unable to work.
- Errors and omissions protects against claims for problems with your product or service that result in a financial loss.
- Business owner’s policy combines protection for all major property and liability risks in one package. This type of policy assembles the basic coverages required by a business owner in one bundle.
- Cyber liability covers loss or theft of data.
Tailor-made Insurance Packages for Small Businesses
Some types of coverage apply to any business, while others are specific to a particular industry.
At Heffernan, we’ve worked hard to provide insurance to businesses working in, but not limited to, the following industries:
And if you don’t see your particular industry or niche listed above, we’re still very happy to research and provide a package that is custom designed just for your needs.
Why You Need Protection
The U.S. Small Business Administration defines small businesses as those with fewer than 500 employees or less than $7 million in total annual receipts. Businesses that fall into this category must prepare for two broad types of liability:
- Internal. This can include everything from on-the-job injuries to discrimination lawsuits. Even if the business is not at fault, legal fees add up quickly.
- External. The possibilities are nearly limitless, from a customer slipping and falling on your property to a client losing money after taking your professional advice.
The frequency and expense of small-business lawsuits can be sobering:

Percentage of small businesses that have been threatened with or involved in a civil lawsuit.
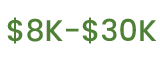
Average legal fees in a workplace-rights case.
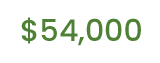
Median cost of a business liability lawsuit.
Heffernan Insurance Brokers for Small Businesses: Your Partner for Success
Meeting the needs of small business owners has remained a passion for Heffernan’s dedicated small business division.
We focus entirely on meeting the specific needs of our small business insurance clients.
Together, we can help keep you on the path to success and scale your benefits as you grow from a basement startup to Fortune 500.
Being a small business does not mean having to settle for small results.
Remember, every large and mid-sized business once was a small business like yours. Let us show you today how easy and affordable peace of mind can be.
 How RSUs Can Impact Work Comp Premiums
How RSUs Can Impact Work Comp Premiums The Factors Behind Rising Commercial Auto Costs and How to Control Them
The Factors Behind Rising Commercial Auto Costs and How to Control Them New California Workplace Violence Protection Plan Requirements
New California Workplace Violence Protection Plan Requirements